Outcomes of Continuation vs Discontinuation of Adalimumab Therapy During Third Trimester of Pregnancy in Inflammatory Bowel Disease - ScienceDirect

IJMS | Free Full-Text | The Role of TNF-α and Anti-TNF-α Agents during Preconception, Pregnancy, and Breastfeeding
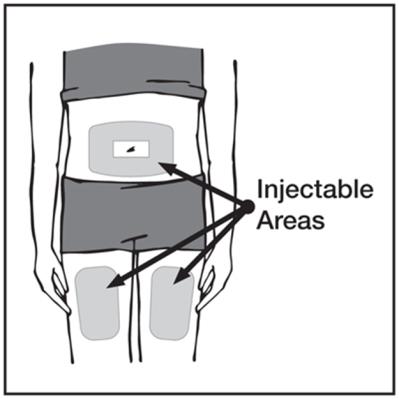
These highlights do not include all the information needed to use HUMIRA safely and effectively. See full prescribing information for HUMIRA. HUMIRA ® (adalimumab) injection, for subcutaneous use Initial U.S. Approval: 2002
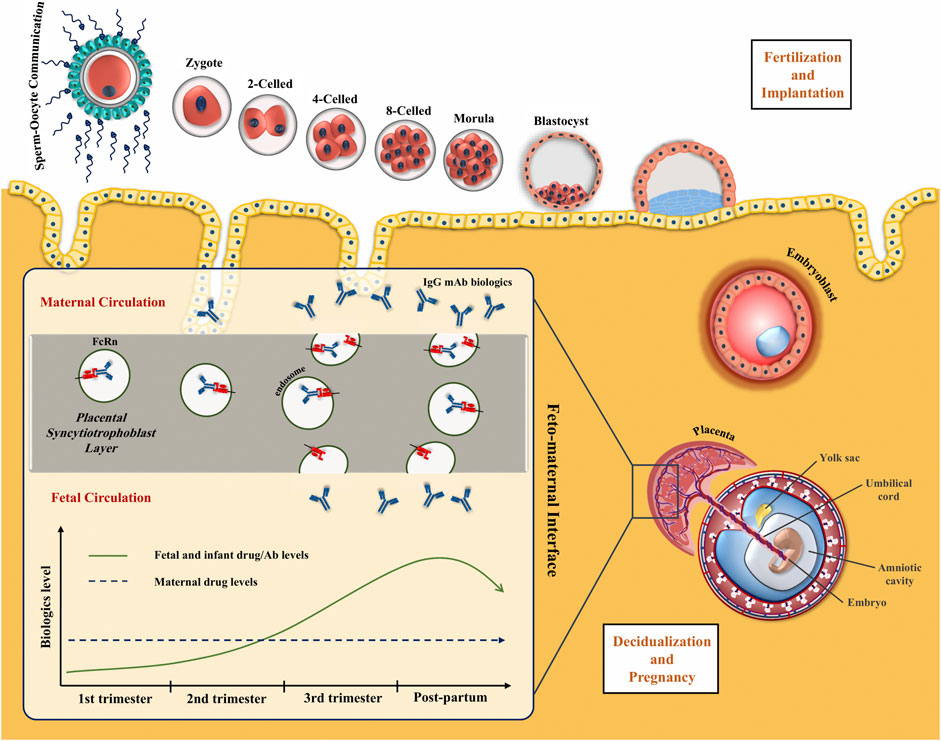
Frontiers | Biologics During Pregnancy and Breastfeeding Among Women With Rheumatic Diseases: Safety Clinical Evidence on the Road

These highlights do not include all the information needed to use HUMIRA safely and effectively. See full prescribing information for HUMIRA. HUMIRA ® (adalimumab) injection, for subcutaneous use Initial U.S. Approval: 2002